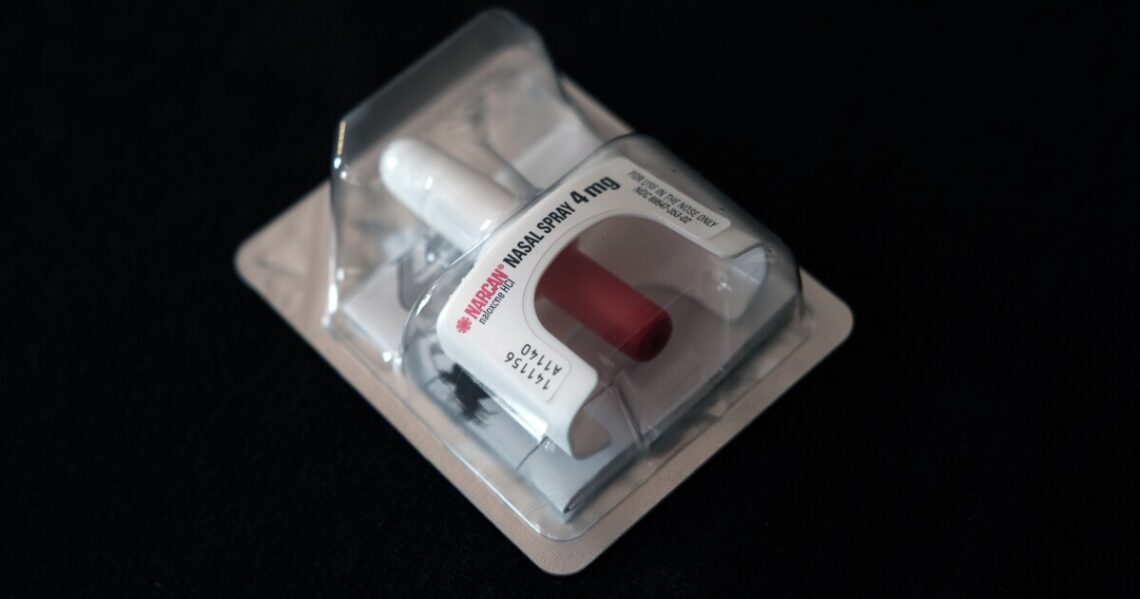
A drug used to reverse opioid overdoses could soon be sold over the counter.
A committee of advisers to the Food and Drug Administration this week voted unanimously in favor of making Narcan, a nasal-spray version of the generic drug naloxone, available without a prescription.
The FDA is weighing the move after fast-tracking an application from Emergent BioSolutions, the maker of Narcan, to let it sell the drug over the counter.
The medication is a key tool in preventing opioid-related overdose deaths,which have climbed in recent years, due in large part to the spread of fentanyl. More than 80,000 people died of overdoses involving opioids in 2021, a historic high, according to the Centers for Disease Control and Prevention.
Experts say naloxone is a safe, effective, easy-to-use medicine that works if administered within the first few minutes of an overdose, with no potential for abuse. The medication works by binding to the same receptors in the brain as opioids, countering their effects.
The advisory panel said doing away with prescriptions for Narcan will save lives.
“For the sake of the public and saving lives, I believe this medication should be available over the counter to the public as soon as possible,” said Dr. Katalin Roth, a professor of medicine at the George Washington University School of Medicine & Health Sciences, after Wednesday’s panel vote.
The FDA plans to make a decision by the end of March.
About the cost
The cost of the life-saving drug could affect whether Narcan going over the counter is indeed accessible
Making Narcan more widely available is an important step in addressing the opioid…
Read the full article here